Sulfur
Atomic Data
| Atomic Number | 16 |
| Symbol | S |
| Atomic Weight | 32.06 u |
| Density (STP) | 2.067 g/cm³ |
| Melting Point | 115.21 °C (388.36 K) |
| Boiling Point | 444.60 °C (717.75 K) |
| Electronegativity | 2.58 (Pauling) |
| Electron Config. | 1s2 2s2 2p6 3s2 3p4 |
| Oxidation States | −2, +2, +4, +6 |
| Phase at STP | Solid |
| Category | Reactive nonmetal |
| Period / Group | 3 / 16 |
| CAS Number | 7704-34-9 |
Electron Configuration
[Ne] 3s2 3p4
| Shell | n | Subshell | Electrons | Cumulative |
|---|---|---|---|---|
| K | 1 | 1s | 2 | 2 |
| L | 2 | 2s | 2 | 4 |
| L | 2 | 2p | 6 | 10 |
| M | 3 | 3s | 2 | 12 |
| M | 3 | 3p | 4 | 16 |
| Total | 16 | 16 | ||
Isotopes of Sulfur
Sulfur has four stable naturally occurring isotopes. ³²S is by far the most abundant at 94.99%; ³6S is present only in trace quantities at 0.01%.
| Isotope | Symbol | Protons | Neutrons | Abundance | Stability |
|---|---|---|---|---|---|
| Sulfur-32 | ³²S | 16 | 16 | 94.99% | Stable |
| Sulfur-33 | ³³S | 16 | 17 | 0.75% | Stable |
| Sulfur-34 | ³⁴S | 16 | 18 | 4.25% | Stable |
| Sulfur-36 | ³⁶S | 16 | 20 | 0.01% | Stable |
Abundance & Occurrence
Sulfur is one of the few elements that occurs in a pure native form, deposited by volcanic activity around fumaroles and in large underground salt-dome reservoirs. It also appears widely as metal sulfide ores (pyrite, galena, cinnabar) and sulfate minerals such as gypsum and barite, and is recovered in large volumes as a refinery byproduct during petroleum desulfurisation.
Earth's Crust (ppm by mass)
Universe (ppm by mass)
Discovery & History
Safety & Handling
- Combustion hazard: Sulfur ignites readily and burns with a pale blue flame, producing sulfur dioxide (SO₂) — a toxic respiratory irritant that causes coughing and eye irritation; at high concentrations it can be life-threatening.
- Dust explosion risk: Fine sulfur powder is flammable and can form explosive dust clouds in enclosed spaces. Grinding or transferring sulfur in bulk requires bonding, earthing, and adequate dust extraction.
- Hydrogen sulfide (H₂S): Many sulfur compounds, particularly H₂S, are acutely toxic. The permissible exposure limit is just 1 ppm. H₂S desensitises the sense of smell at higher concentrations, removing the natural warning signal.
- Low acute toxicity in bulk: Solid elemental sulfur poses minimal hazard from skin contact or small ingestion and is used in approved garden fungicide products. Gloves and eye protection are sufficient for routine handling.
- Storage: Store away from oxidising agents, open flames, and heat. Keep in cool, dry, well-ventilated areas in closed containers to minimise dust generation and moisture absorption.
Sulfur in the Real World

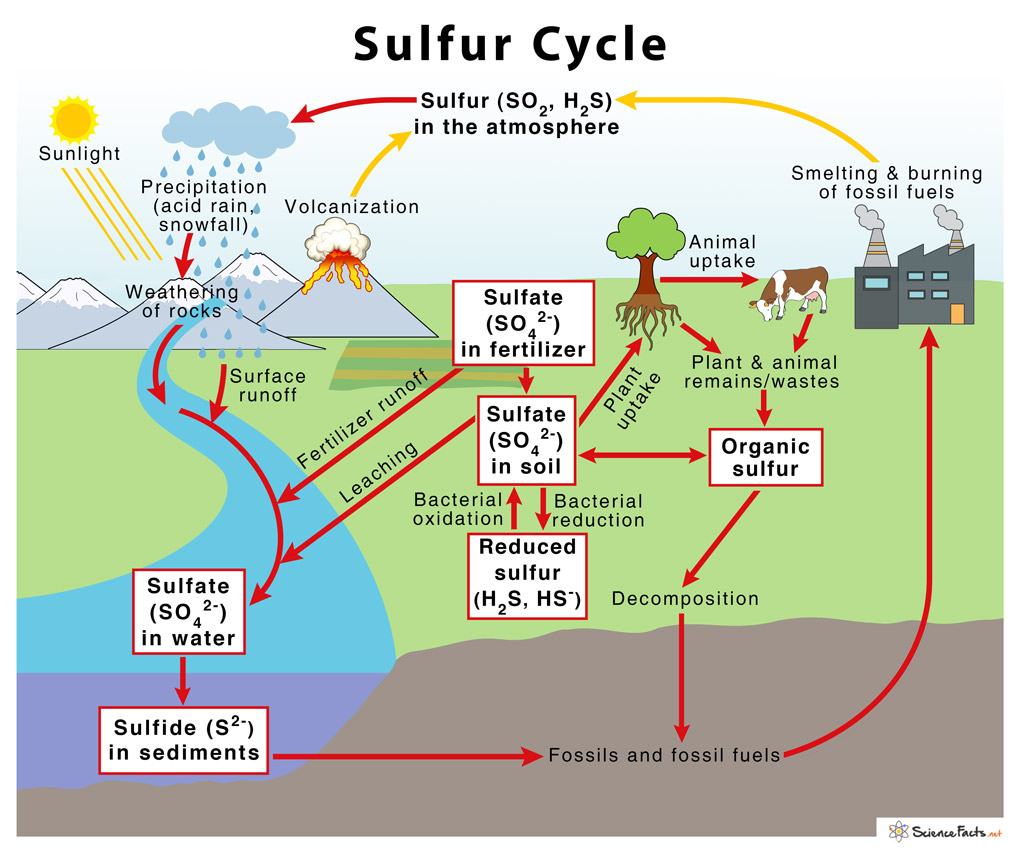

Real-World Uses
- Sulfuric acid production — Around 85% of all mined sulfur is converted into sulfuric acid (H₂SO₄) via the contact process; sulfuric acid is the world’s most-produced industrial chemical, central to fertiliser manufacturing, metal refining, battery electrolytes, and chemical synthesis.
- Rubber vulcanisation — Sulfur cross-links polymer chains in natural and synthetic rubber, dramatically improving strength, elasticity, and resistance to heat and chemicals; used in tyres, seals, conveyor belts, and hoses.
- Fungicide and pesticide — Elemental sulfur is one of the oldest known pesticides, still widely used in agriculture to control powdery mildew and other fungal diseases on crops; it is approved for organic farming.
- Pharmaceuticals — Sulfur is an essential component of the amino acids cysteine and methionine, and appears in many drugs including sulfonamide antibiotics; topical sulfur preparations treat acne, rosacea, and seborrhoeic dermatitis.
- Gunpowder — Traditional black powder contains around 10% sulfur alongside charcoal and potassium nitrate; sulfur lowers the ignition temperature of the mixture and stabilises its combustion.
Downloadable Resources
Free periodic table reference sheets for classrooms, study sessions, and laboratory use.
Frequently Asked Questions
What is sulfur used for?
Sulfur's biggest use is in making sulfuric acid, the world's most-produced industrial chemical, which is essential for fertiliser manufacture, metal processing, and chemical synthesis. Sulfur is also used in rubber vulcanisation, fungicides and pesticides, pharmaceuticals, and gunpowder.
Is sulfur dangerous?
Elemental sulfur in bulk form has low acute toxicity and is not considered dangerous to handle. The main hazard arises when it burns: combustion produces sulfur dioxide (SO₂), a toxic, choking gas that irritates the lungs and contributes to acid rain. Hydrogen sulfide (H₂S), a common sulfur compound, is highly toxic even at low concentrations.
Why does sulfur smell like rotten eggs?
Pure elemental sulfur is actually nearly odourless. The rotten-egg smell commonly associated with sulfur comes from hydrogen sulfide (H₂S) and organic sulfur compounds such as mercaptans, which are produced by the bacterial decomposition of sulfur-containing proteins.
Where is sulfur found in nature?
Sulfur occurs naturally as a pure element near volcanic vents and hot springs, and in large underground deposits — historically mined by the Frasch process. It is also found in metal sulfide ores such as iron pyrite (FeS₂), and is recovered in large quantities as a byproduct of petroleum and natural gas processing.